Matthew Feshbach, Founder & CEO
 Matt has taken an unconventional path throughout his business career. He did not attend college but instead taught tennis at private homes in Silicon Valley. He developed professional and personal relationships with some of the most prominent leaders in the San Francisco investment community through that experience. They inspired him to study securities analysis and begin investing himself in 1979.
Matt has taken an unconventional path throughout his business career. He did not attend college but instead taught tennis at private homes in Silicon Valley. He developed professional and personal relationships with some of the most prominent leaders in the San Francisco investment community through that experience. They inspired him to study securities analysis and begin investing himself in 1979.
Personal investing led to the co-founding, with his brothers Kurt and Joe, of Feshbach Bros in 1982. Feshbach Bros. managed the first “short-only” hedge fund. Assets under management grew from $20,000 to $1 Billion in 9 years. According to a survey by Piper, Jaffrey, Feshbach Bros achieved the top investment record of any money manager, equity or debt, from 1985- 1990. Matt’s investment career has now spanned over 40 years through the stock market’s ups and downs.
In 2009, he and his brother, Joe, began researching adult stem cells and their potential due to their interest in a publicly-traded company. They spoke to researchers from all over the world – Israel to Japan, Holland to Spain, New York to Los Angeles, Chicago to Dallas, and so on. Their quest was to understand the best source of adult stem cells, as well as the science, technologies, and unmet medical needs that cell therapy potentially address.
As a result of their collaboration, they came to a well-informed belief that our fat (adipose tissue) is the most accessible, abundant, and potent source of adult stem cells. The stem cells in our fatty tissue are called adipose stem cells (ASCs). Further, in our fat, is a mixed population of cells. This diverse population is called adipose-derived stem and regenerative cells (ADRCs).
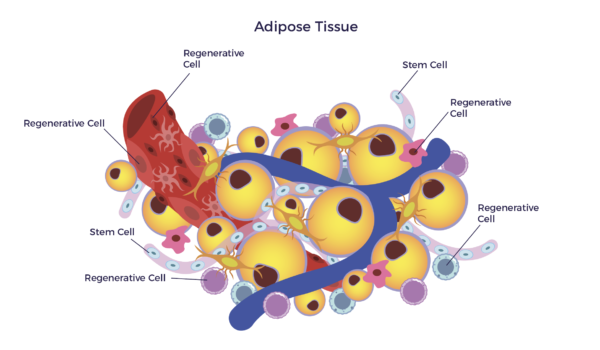
Even in those early days, the science suggested that ADRCs were safe and potentially effective for a broad spectrum of chronic diseases.
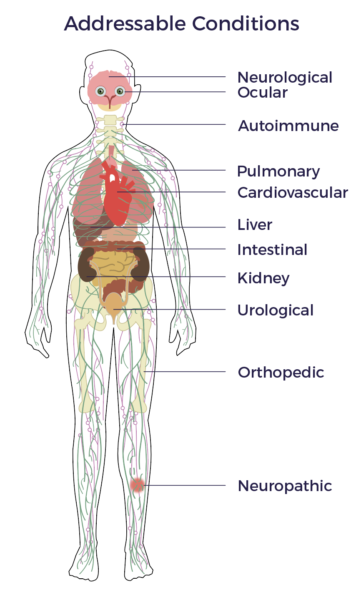
Researchers and investigators have made significant progress since Matt first began to learn about ADRCs. As of December 2019, a PubMed search of “adipose stem cells” yields over 65,000 papers. This vast and growing body of literature (including hundreds of published clinical studies) strongly supports the potential that he perceived back in the early days of his research into the field.
Along with his brother Joe, Matt founded Okyanos (Oh-key-ah-nos) Cell Therapy in August 2011 in Freeport, Bahamas. The Greek God of Rivers, “Okyanos,” symbolizes the restoration of blood flow, an essential mechanism of action of ADRCs. Shortly after helping to create Okyanos, Joe sadly died of a cardiac arrest. Joe’s death prompted Matt to be even more dedicated to his purpose to help patients with chronic disease with their own adult stem cells.
Working with the Bahama’s Minister of Health, Perry Gomez, MD, Matt drafted the framework for what developed into the Bahamas Stem Cell Research and Therapy Act while concurrently building and staffing Okyanos. Due to Matt’s uncompromising standards, Okyanos came to be known as the first purpose-built cell therapy center of excellence in the world.
During Matt’s tenure, Okyanos treated more than 300 patients with IV therapy of ADRCs, often combined with direct injections of micro-fat to address localized conditions involving bones, muscles, nerves, wounds, and scars. There were no reported cell-related adverse events, and >85% of patients reported improved symptoms, function, and quality of life. Matt left Okyanos in May 2017, taking with him his purpose to help people suffering from chronic degenerative diseases live better lives.
On May 30, 2018, an enlightened and extraordinary humanitarian bill, the Federal Trickett Wendler, Frank Mongiello, Jordan McLinn, and Matthew Bellina Right to Try Act of 2017 (Right to Try Act), was signed into law by President Trump.
Fortuitously, the Right to Try Act’s requirements dovetailed with the technologies, protocols, and patient criteria that Matt believed would best address the unmet need of patients suffering from life-threatening and debilitating chronic degenerative diseases. And so, Matt established AMBROSE Cell Therapy in August 2018.
Today, as the CEO of AMBROSE, Matt is pursuing his overarching purpose to help people with complex chronic diseases live better lives. AMBROSE provides the “right to try” cell therapy in Beverly Hills, CA under the Right to Try Act. And, in complete alignment with our Mission: To help people with chronic degenerative diseases improve symptoms, function, and quality of life using adult stem cell therapy.
In sum, Matthew Feshbach has combined his four decades of investment experience with his purpose to help others. He is an individual with no formal higher education, much less medical training. Yet, physicians, stem cell researchers, and patients respect him for his impressive knowledge of the science underpinning adult stem cell therapy’s potential effectiveness. Perhaps most important, from the day he began his journey he has aspired and worked diligently to bring the highest standard of cell therapy care to help patients who need it most.
