Biodistribution of Adult Stem Cells
Mesenchymal Stem Cells
In 1991, Professor Arnold Caplan published the seminal paper, Mesenchymal Stem Cells (MSCs). In the article, he described MSCs as having the ability to differentiate and self-renew into multiple tissue types, as well as neurons and blood vessels. [1] In other words, they are ‘multipotent,’ meaning they can produce more than one type of specialized cell of the body, but not all types. Differentiation and self-renewal are the unique characteristics required of a cell to be a stem cell.
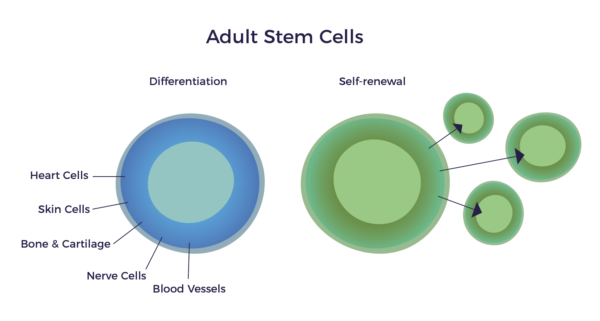
In the article’s introduction, he called MSCs, “the basis for the emergence of a new therapeutic technology of self-cell repair” (emphasis added). His tireless research gained extensive support from hundreds of other scientists. In fact, according to a PubMed search, there are now over 160,000 published papers that discuss MSCs. This vast body of literature has led to a broad agreement as to how these repair cells migrate to and repair diseased tissues and organs. The science has also progressed to include an understanding as to how MSCs restore normal function in our vascular, immune, and endocrine (hormone) systems.
Research has further progressed to include an appreciation of how MSCs do their job (and has led to a proposed new name for them). More on the new name later; first, here is the back story.
Biodistribution
Since the early days of research into MSCs, scientists have been on a quest to determine the optimum delivery route. Much like planning the most efficient course through a labyrinth-like city (such as London), they want our stem cells to stay clear of blind alleys, wrong-way streets, and circuitous routes. The investigators also recognize some courses may be riskier than others – even if seemingly more efficient per the map, e.g., directly injecting stem cells into the brain or the eye.
But there is a new angle to the planning process that is increasingly appreciated by researchers: The underlying role that low-level systemic inflammation (inflammaging) plays in chronic disease and the potential for stem cells to calm that down. More on inflammaging later; first, how do MSCs do their job?
Mechanisms of Action
Mechanism of action (MOA) refers to the way our stem cells (or drugs) produce their effect. In the case of MSCs, their MOAs are analogous to the fire department: They respond to a fire alarm – i.e., an inflammatory signal – and beeline to the source of it. MSCs have been called “guardians of inflammation.” [2] Once they get to the scene of the fire, they release hundreds of bioactive signaling molecules called “cytokines.” The cytokines activate the local cells in the neighborhood to do their jobs. This cell-to-cell communication is called the paracrine effect.
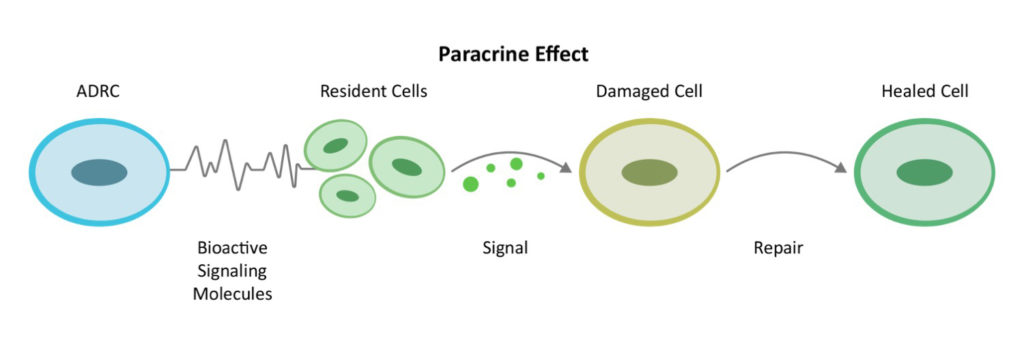
In short, since Dr. Caplan’s groundbreaking paper was published, researchers have verified that homing and the paracrine effect are the central roles of MSCs in the repair process. And so, that is why Prof. Caplan proposed changing the name to “Medicinal Signaling Cells.” [3]
Biodistribution Studies
Biodistribution studies have established that adult stem cells delivered IV migrate through the lungs and “park” there (“first-pass effect”) – and then move on. (There is some confusion as to whether the first-pass effect is a “dead-end street,” but suffice to say, research has established that it is not the case.) [4] [5] [6]
As they continue on their mission to fight inflammation and repair the body, they journey through the vascular system and organs to the spleen, our largest immune organ. They then route through the lymphatic system.
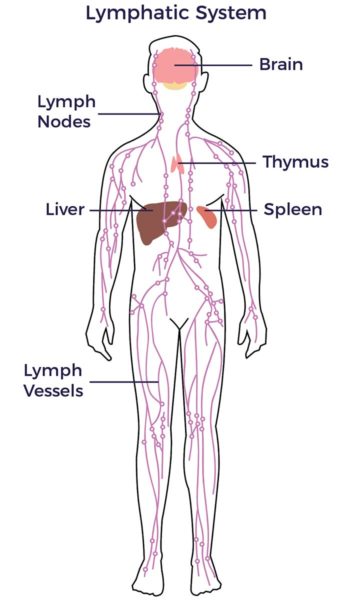
The lymphatic system, which runs parallel to the vascular system, is an essential part of the immune system. It carries lymph (a clear fluid containing a high number of a type of white blood cells called lymphocytes. Lymphocytes fight infection and destroy damaged or abnormal cells. They are abundant in the mixed population of cells accessed from our adipose tissue for cellular therapies.
Biodistribution Studies
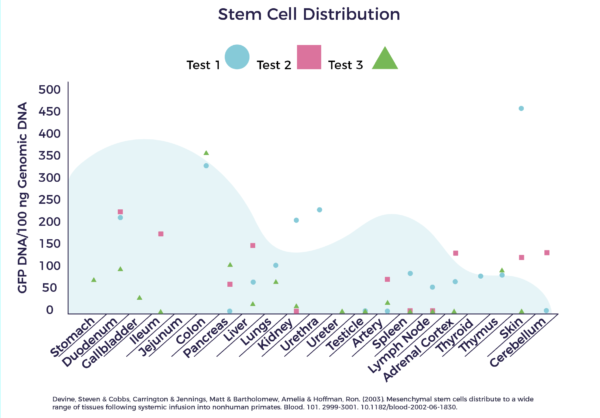
A well-designed study of IV infusion of adult stem cells in baboons tracked the migration. The graph below shows distribution to all of the major internal body organs. [7]
Add to that, in a cardiac cell therapy study, researchers at Texas Heart Institute and MD Anderson tracked stem cells injected in the hearts of pigs. They also found the MSCs traveled through the blood vessel system into the spleen and then continued to move through the lymphatics. [8]
As a side note, lest we discount the potential of self-cell repair, a study in mice found that systemically delivered adult stem cells could repair chemically-induced corneal damage – without there being any of the MSCs directly applied to the cornea. [9]
Summary
For more on why AMBROSE accesses the MSCs and other regenerative cells from your adipose tissue, please read Why Adipose Tissue. Connecting this back to our Areas of Focus and Our Protocol, ADRCs, delivered IV, have the potential to home to all of the major organs and initiate a process of repair.
[2] DJ Prockop and JY Oh MSCs as Guardians of Inflammation Molecular Therapy Vol. 20 no. 1, 14–20 Jan. 2012
[3]A Caplan Mesenchymal Stem Cells: Time to Change the Name! Stem Cells Translational Medicine 2017;6:1445–1451
[4]Pulmonary Passage is a Major Obstacle for Intravenous Stem Cell Delivery: The Pulmonary First-Pass Effect
[5]AS Cornellisen et al Organ-specific migration of mesenchymal stromal cells: Who, when, where and why? Immunology Letters 168 (2015) 159–169
[6]M El-Ensary et al Mesenchymal stem cells are a rescue approach for recovery of deteriorating kidney function Nephrology 17 (2012) 650–657
[7]S. Devine et al Mesenchymal stem cells distribute to a wide range of tissues following systemic
infusion into nonhuman primates Blood 15 April 2003 Volume 101, Number 8 2999
[8]EC Perin et al. (2011) Imaging Long-Term Fate of Intramyocardially Implanted Mesenchymal Stem Cells in a Porcine Myocardial Infarction Model. PLoS ONE 6(9): e22949.
[9]G Roddy et al Action at a Distance: Systemically Administered Adult Stem/Progenitor Cells (MSCs) Reduce Inflammatory Damage to the Cornea Without Engraftment and Primarily by Secretion of TNF-a Stimulated Gene/Protein 6 STEM CELLS 2011;29:1572–1579
